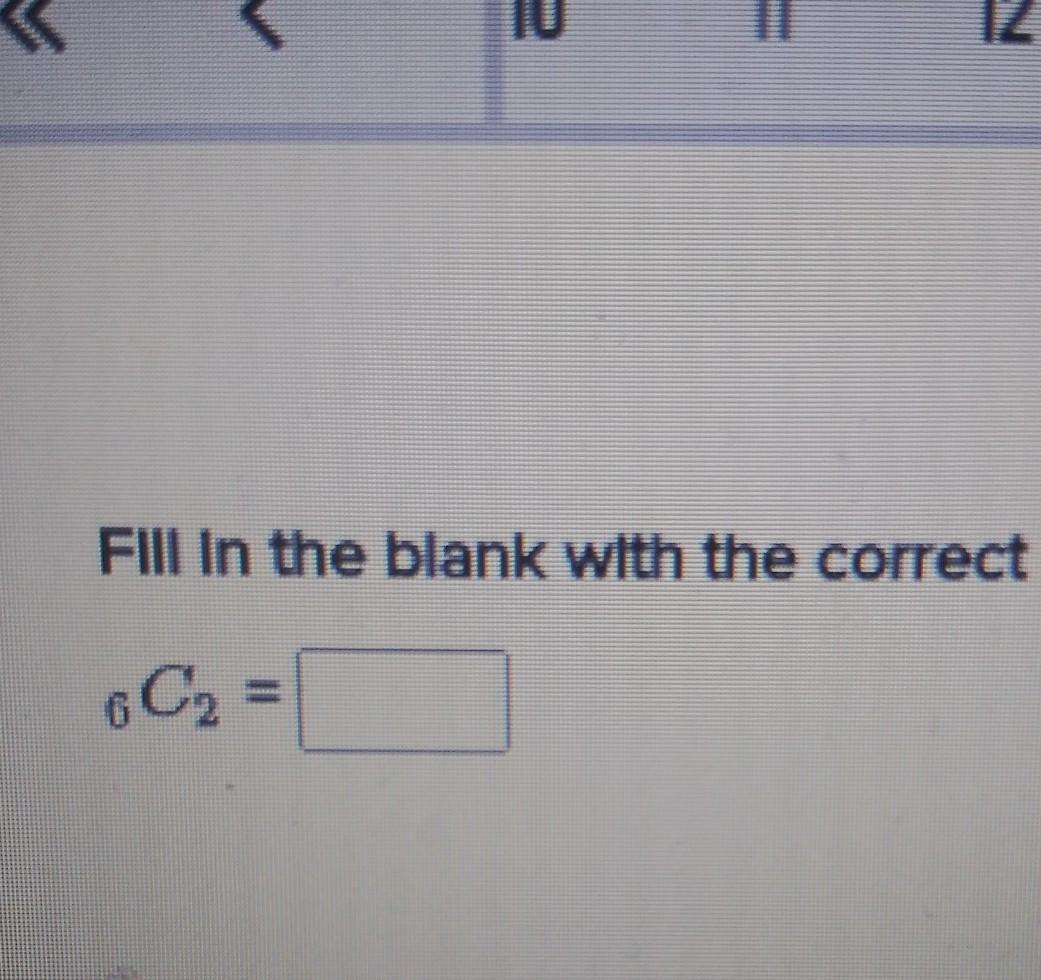
The main principles of humanistic theory applied to social work include the following EXCEPT O All individuals experience and understand their changing world in unique ways O Individuals seek congruence between one's self-concept and their experiences O A person's behavior is influenced by one's perceptions of their world O Obstacles to change are a function of one's ego defenses
-
Subject:
Social Studies -
Author:
onésimowarner -
Created:
1 year ago
Answers 1
AI generated Answer
Answer: Obstacles to change are a function of one's ego defenses
Explanation: Humanistic theory emphasizes valuing the unique individual, their perceptions, and the search for self-fulfillment. It focuses on helping individuals find the positive in themselves and in their situation, emphasizing self-efficacy and personal growth. While ego defenses do play a role in behavior as part of the psychodynamic approach, this is not a principle of humanistic theory as it implies that outward influences can affect the individual in a way where they cannot control their behavior.
-
Rate an answer:
0
Do you know an answer? Add it here!
Unable to find an answer to your question?
Don't worry! There are several alternative approaches you can try to resolve your query. Here are some tips to help you find answers in different ways:
- Reframe your question: Sometimes, the way you phrase your question can limit your search results. Try rephrasing it using different keywords or providing more context to get better results.
- Utilize social media: Post your question on social media platforms, particularly those focused on professional or specialized topics. Twitter, LinkedIn, and Facebook groups can connect you with individuals who may have relevant expertise or experiences to share.
- Consult subject matter experts: Reach out to experts in the field related to your question. Many professionals are willing to help and share their knowledge. You can connect with them through email, LinkedIn messages, or by attending relevant conferences or events.
- Use our website to find your question through the search box above, or you can sign up to ask your question for our big educational community. Our experts will review your question, and you will get a quick and quality answer.
- Collaborate with others: If your question is related to a specific project or problem, consider collaborating with others who might have complementary skills or knowledge. Teamwork can lead to innovative solutions and shared insights.
Remember, the process of finding answers often involves persistence, creativity, and an open mind. By exploring various resources, reaching out to others, and being proactive in your search, you increase your chances of finding the information you need. Happy quest for knowledge!
Choose a language and a region
How much to ban the user?
1 hour
1 day
100 years